Sunisam 50 Mg Sunitinib
Price 1100 INR/ Strip
Sunisam 50 Mg Sunitinib Specification
- Dosage Form
- Capsulas
- Indication
- Renal cell carcinoma, Gastrointestinal stromal tumor (GIST), Pancreatic neuroendocrine tumors
- Salt Composition
- Sunitinib Malate 50 mg
- Origin
- India
- Feature
- Oral multi-targeted receptor tyrosine kinase inhibitor
- Ingredients
- Other
- Application
- Anticancer medication
- Physical Color/Texture
- Yellow powder-filled hard gelatin capsules
- Storage Instructions
- Keep dry & cool place
- Shelf Life
- 24 months
- Country of Import
- India
- Route of Administration
- Oral
- Strength
- 50 mg per capsule
- Regulatory Status
- Approved Rx
- Prescription/Non Prescription
- Prescription only
- Therapeutic Class
- Antineoplastic Agents
- Contraindications
- Known hypersensitivity to sunitinib or any component of the formulation
- Manufactured By
- Samarth Life Sciences Pvt. Ltd.
- Packaging Size
- 28 capsules per box
- Marketed By
- Sunisam
- Warning/Precaution
- To be used under medical supervision
Sunisam 50 Mg Sunitinib Trade Information
- Minimum Order Quantity
- 4 Strips
- Supply Ability
- 1000 Strips Per Month
- Delivery Time
- 7 Days
About Sunisam 50 Mg Sunitinib
Key Features & Application: Sunisam 50 Mg Sunitinib
Sunisam 50 Mg Sunitinib is an orally administered multi-targeted receptor tyrosine kinase inhibitor, indicated for various cancers such as renal cell carcinoma, GIST, and pancreatic neuroendocrine tumors. Designed for use by medical professionals and patients under strict medical guidance, Sunisam's application is straightforward, thanks to its capsule form. Its innovative action targets cancer pathways, delivering exceptional support in oncology care. The medication is primarily used in clinics, hospitals, and specialized medical centers.
Domestic Market, Certification, and Logistics: Sunisam 50 Mg Sunitinib
Sunisam 50 Mg Sunitinib commands significant market value in India, serving trusted healthcare institutions and pharmacies nationwide. Goods transport is efficiently managed to ensure the product's integrity from manufacturer to end-user, adhering to regulatory compliance. Certified for prescription-based distribution, samples are provided based on formal policy requirements. The handover process prioritizes safe, verified delivery backed by documentation, ensuring only authorized medical professionals access this critical anticancer medication.
FAQ's of Sunisam 50 Mg Sunitinib:
Q: How should Sunisam 50 Mg Sunitinib be administered?
A: Sunisam 50 Mg Sunitinib is intended for oral use and should be taken exactly as prescribed by your oncologist or qualified healthcare provider. Do not alter the dosage or discontinue use unless directed.Q: What are the primary benefits of using Sunisam 50 Mg Sunitinib?
A: Sunisam 50 Mg Sunitinib is designed to inhibit cancer cell growth in conditions such as renal cell carcinoma, GIST, and pancreatic neuroendocrine tumors, improving treatment outcomes when used under proper medical supervision.Q: When should Sunisam 50 Mg Sunitinib not be used?
A: Sunisam 50 Mg Sunitinib should not be used if you have a known hypersensitivity to sunitinib or any of its components. Always inform your doctor of allergies or adverse reactions prior to starting therapy.Q: What precautions must be taken during the usage of Sunisam 50 Mg Sunitinib?
A: This medication should be used strictly under medical supervision, with regular follow-up for monitoring potential side effects. Store the capsules in a dry, cool environment and keep them out of reach of children.Q: Where is Sunisam 50 Mg Sunitinib manufactured and marketed?
A: Sunisam 50 Mg Sunitinib is manufactured by Samarth Life Sciences Pvt. Ltd. and marketed by Sunisam, both based in India, and is available via authorized distributors, suppliers, and traders throughout the country.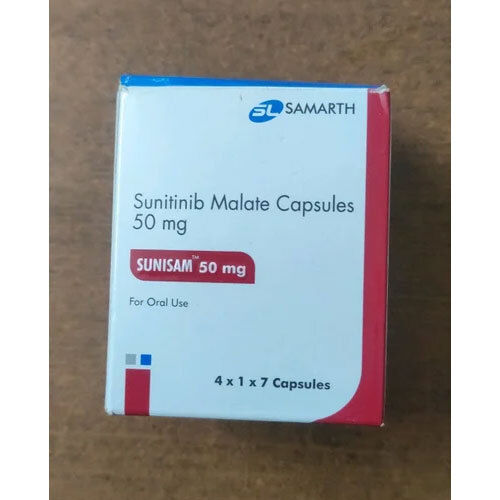

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti cancer Medicines Category
Evermil 10 Mg
Price 4750 INR / Strip
Minimum Order Quantity : 2 Strips
Dosage Form : Tablets
Storage Instructions : Keep dry & cool place
Feature : Other, High efficacy as an mTOR inhibitor, targeted cancer therapy
Physical Color/Texture : Other , White to offwhite, round shaped tablets
Lenva 4 Mg
Price 950 INR / Bottle
Minimum Order Quantity : 6 Bottles
Dosage Form : Capsules
Storage Instructions : Keep dry & cool place
Feature : Other, Oral administration, targeted therapy
Physical Color/Texture : Other , Hard gelatin capsule, blue opaque cap / white opaque body
Pazotab 400 mg
Price 2700 INR / Bottle
Minimum Order Quantity : 10 Bottles
Dosage Form : Tablets
Storage Instructions : Keep dry & cool place
Feature : Other, Oral administration, prescription only
Physical Color/Texture : Other , Yellow, filmcoated tablets
Unigef 250mg Tablet
Price 350 INR / Strip
Minimum Order Quantity : 10 Strips
Dosage Form : Tablets
Storage Instructions : Keep dry & cool place
Feature : Other, Filmcoated, easy to swallow, precise dosing
Physical Color/Texture : Other , White to offwhite, filmcoated tablet

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS
